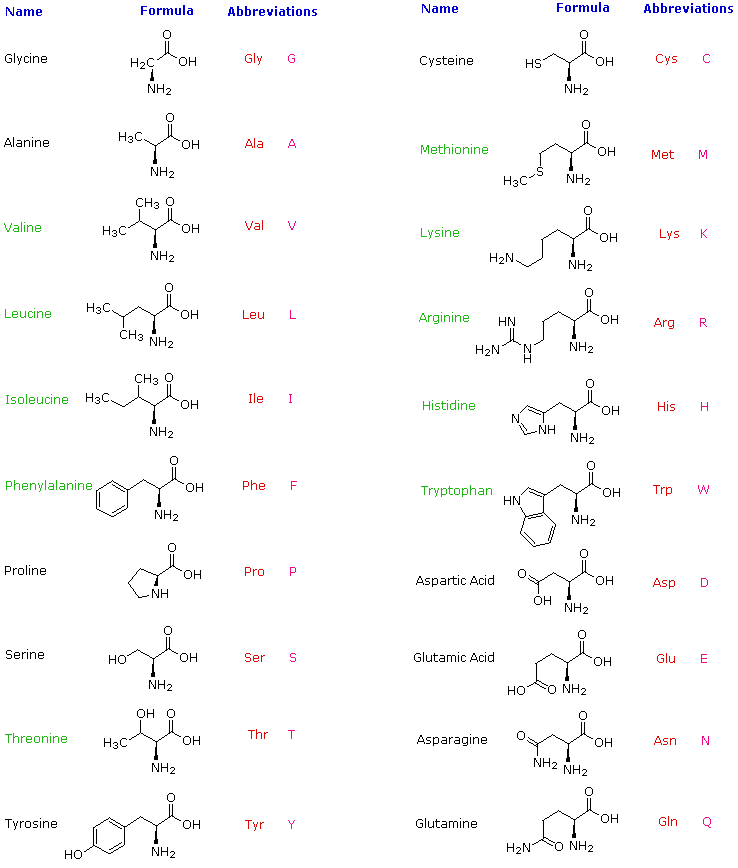
In protein molecules, these residues are often found close to the interface between a protein and solvent. The aromatic amino acids tryptophan (Trp, W) and Tyr and the non-aromatic methionine (Met, M) are sometimes called amphipathic due to their ability to have both polar and nonpolar character. This ability makes histidine useful in enzyme active sites when the chemical reaction requires proton extraction. Within protein molecules, the pKa may be modulated by the environment so that the side chain may give away a proton and become neutral or accept a proton, becoming charged. When both groups are protonated, the side chain has a charge of +1. It has two –NH groups with a pKa value of around 6. Histidine (His, H), on the other hand, depending on the environment and pH, can be polar or carry a charge. 
Asparagine (Asn, N) and glutamine (Gln, Q) also belong to this group and may donate or accept a hydrogen bond. Tyrosine is also involved in metal binding in many enzymatic sites. Furthermore, this group can form a hydrogen bond with another polar group by donating or accepting a proton (a table showing donors and acceptors in polar and charged amino acid side chains can be found at the FoldIt site. For example, serine (Ser, S), threonine (Thr, T), and tyrosine (Tyr, Y) are polar since they carry a hydroxylic (-OH) group. When considering polarity, some amino acids are straightforward to define as polar, while in other cases, we may encounter disagreements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2023
Categories |
 RSS Feed
RSS Feed
